Professional, Customizable Arabidopsis thaliana Transformation Services
Lifeasible specializes in providing efficient Arabidopsis transformation services to support your research in functional genomics, gene editing, and trait improvement. Our service platform offers seamless support from vector design to stable strain construction, providing tailored solutions for academic and industrial clients dedicated to Arabidopsis research.
TARGET ECOTYPES
Col-0, Ler, Ws,
& Mutant backgrounds
EXPECTED YIELD
10–50
Independent T1 Arabidopsis lines
EDITING EFFICIENCY
Up to 80%
High-efficiency gene editing
LEAD TIME
4–6 weeks
From dip to T1 seeds
Standard Package
Efficiency Focused
Premium Package
Full-Service Custody
Stable transformation is central to Arabidopsis research, enabling researchers to generate heritable transgenic lines for long-term genetic studies. Our service is based on the widely used Agrobacterium-mediated floral dip method, which has been refined over decades for high reproducibility and efficiency.
Unlike tissue culture-based methods, the floral dip method requires no callus induction or regeneration, significantly simplifying the process and reducing the risk of somaclonal variation. This method allows for rapid generation of transgenic seeds (T1) in just weeks, enabling rapid downstream analysis.
![]()
Agrobacterium with T-DNA Vector
Carries the gene of interest inside a Ti plasmid (engineered)
![]()
Contact with Arabidopsis Flowers (Floral Dip)
Agrobacterium cells adhere to ovule tissues
![]()
T-DNA Transfer into Plant Cells
Bacterial virulence proteins mediate DNA transfer
![]()
T-DNA Integration into Plant Genome
Stable insertion into nuclear DNA
![]()
Transgenic Seeds (T1 Generation)
Seeds carry the new trait for heritable expression
For researchers who need rapid results but don't want to invest the time required to establish stable lines, our transient transformation services offer a powerful alternative. These systems enable transient gene expression and are ideal for exploratory studies, rapid functional analysis, or preliminary data collection before stable transformation. We offer a variety of transient expression protocols to meet diverse research needs.
![]()
DNA Construct Preparation
Plasmid carrying gene of interest
![]()
DNA Delivery into Plant Cells
![]()
Expression Without Genomic Integration
DNA remains episomal or transiently active
![]()
Rapid Protein / Reporter Gene Expression
Detected within 1–7 days, but not heritable
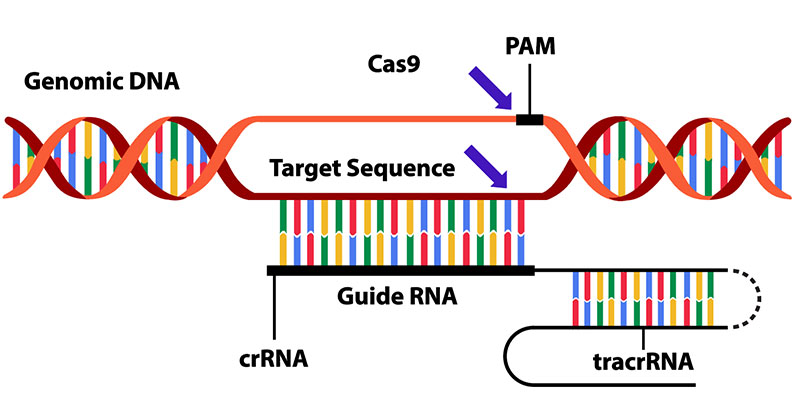
Our CRISPR/Cas9-based transformation services provide a precise and efficient toolkit for genome editing in Arabidopsis. We offer end-to-end support, from sgRNA design to delivery of edited lines.
CRISPR/Cas9 services can be combined with stable and transient expression systems, providing flexibility tailored to your timeline and project scope.
Gene function is often best understood by enhancing or silencing its activity. To support these experiments, we offer custom overexpression and RNAi constructs to enable gain- or loss-of-function studies.
This service ensures that customers receive fully validated, transformation-ready constructs with the option of integration into stable or transient systems.
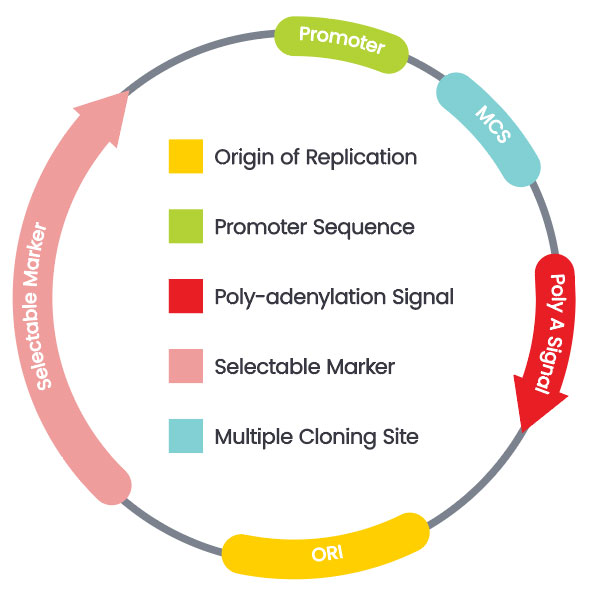
Our molecular biology team designs and constructs custom vectors for Arabidopsis transformation projects. We can use your provided vectors or build custom constructs from scratch.
Service Capabilities
Start Your Arabidopsis Project Today
Standard Deliverables
Optional Upgrades

We offer a variety of support services to meet your unique research needs:
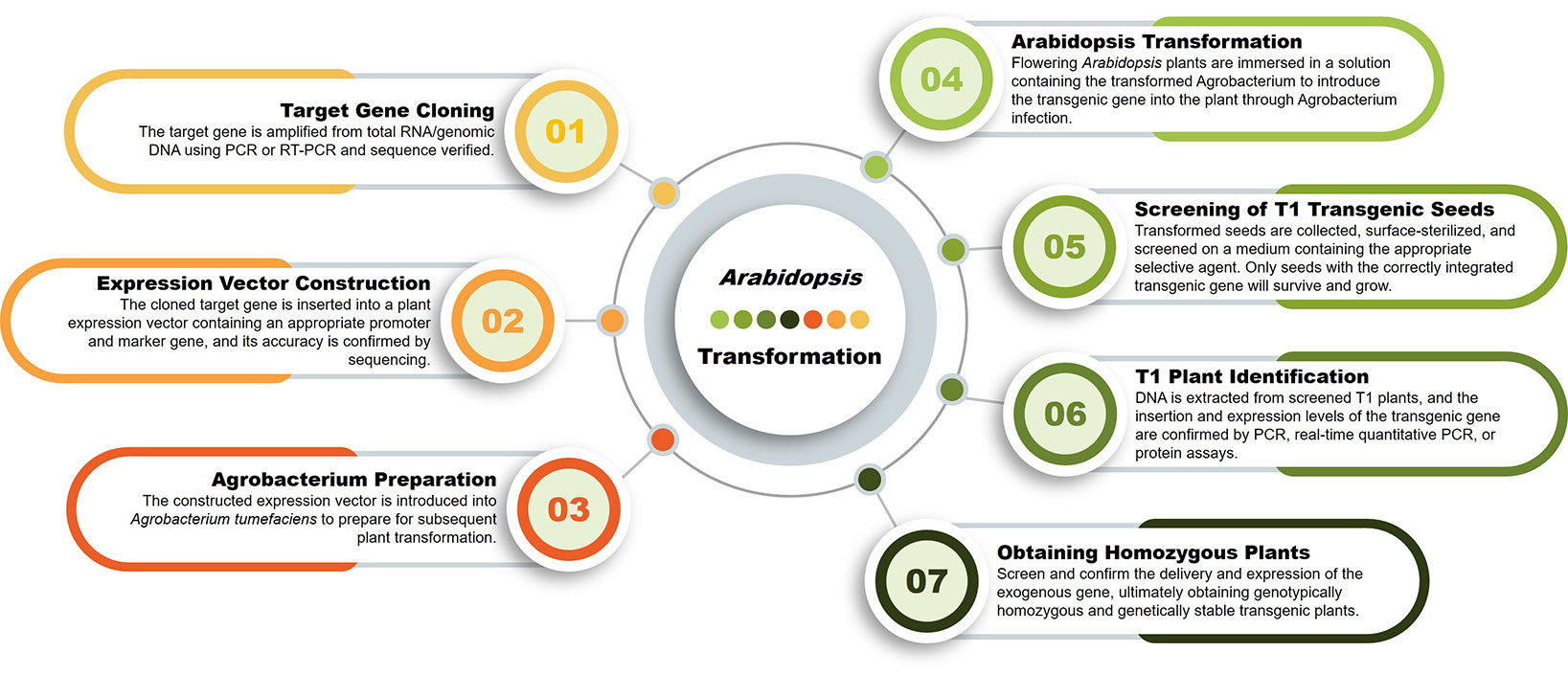
Vector Construction
Agrobacterium Preparation
Floral Dip Transformation
T1 Seed Harvesting
T1 Seedling Screening and PCR
T2/T3 Generation (optional)
We ensure regular updates and project flexibility based on your goals and schedule.
Multi-Plasmid Co-Transformation in Arabidopsis
Successfully conducted co-transformation across seven distinct plasmid combinations. Utilizing our optimized floral dip protocol, we achieved high integration efficiency, securing multiple positive lines with confirmed stable inheritance in T2 generations.

Stable Expression in A. thaliana and B. napus L.
We successfully established stable expression in both Arabidopsis and oilseed rape (B. napus) using Agrobacterium-mediated transformation. The integration of target genes in T1/T2 seedlings was rigorously confirmed through both genomic PCR and Western Blot analysis. This dual-species validation showcases our platform's versatility in Brassicaceae research, with ongoing optimization of protein tags to further enhance expression levels for downstream functional proteomics.
High-Efficiency Arabidopsis thaliana Transformation
Recent advancements in A. thaliana research, as detailed in Nature Protocols, demonstrate that the integration of SaCas9 with egg-cell-specific promoters significantly enhances the efficiency of heritable genomic modifications. This methodology enables precise double-strand breaks and complex chromosomal engineering, such as Mb-sized inversions and translocations, within the plant somatic cells.
Empowering researchers and agricultural leaders with reliable transformation solutions
"The efficiency of the Arabidopsis thaliana Transformation Service is remarkable. We received our transformed lines with precisely the modifications we requested, significantly accelerating our trait development pipeline in the AgBio sector.”
Dr. Richardson
Senior Scientist
USA
"From vector construction to validated T3 homozygous Arabidopsis thaliana lines, the end-to-end support was seamless. The bi-weekly updates from the project managers kept us well-informed throughout the entire transformation process.”
Dr. Thompson
Lab Manager
USA
"We were struggling with transformation efficiency in-house. Lifeasible’s Arabidopsis thaliana Transformation Service achieved nearly 80% success rate on our complex target sites, proving to be a major time-saver for our gene editing projects.”
Dr. Jenkins
Senior Researcher
UK
"The guaranteed delivery of 10–20 independent T1 Arabidopsis thaliana lines provided us with unprecedented biological replicates for our downstream phenotyping. The consistency and quality of the transformation were exceptional, reflecting the professional service offered.”
Dr. Müller
Principal Investigator
Germany
Expertise & Capacity
Our facility processes 1,000+ constructs annually with zero queuing time, managed by senior scientists experienced in A. thaliana genetics.
High Success Rate
Optimized floral dip protocols ensure a 98%+ success rate and reliable delivery of ≥10-20 independent T1 lines per construct.
Project Transparency
Dedicated Ph.D. project managers provide bi-weekly updates and comprehensive molecular validation reports for full status visibility.
End-to-End Solutions
From gene synthesis and mutant background selection to validated T3 homozygous lines, we provide seamless support for every step.
Ready to start your Arabidopsis thaliana transformation project?
Contact us for a free consultation, a custom quote, or to discuss your project needs with a technical expert.
Arabidopsis thaliana, also known as mouse-ear cress, is the premier model organism in plant biology. Its relatively small genome (135 Mbp), short life cycle, and exceptional adaptability to various environments—including growth rooms, chambers, greenhouses, and even window ledges—make it an ideal biological system for studying plant genetics, evolution, and development.
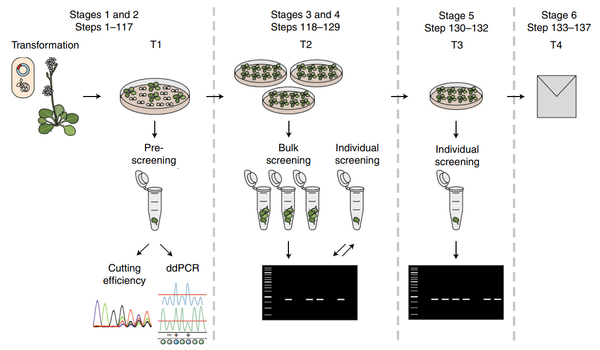
The most widely used method for generating stable transgenic Arabidopsis thaliana is the Agrobacterium-mediated floral dip. During this process:
Compared to tissue culture-based methods, the floral dip requires minimal labor and simple equipment while providing high-efficiency results.
Our Arabidopsis thaliana transformation platform supports a broad spectrum of research goals, from fundamental genetics to translational agricultural science:
You can provide either the plasmid vector or the gene sequence. If you only provide the sequence, we will handle the cloning. Please provide us with the promoter type, selection marker, and target ecotype.
We typically achieve transformation efficiencies of 0.5-2%, generating multiple independent lines for each construct—sufficient for reliable downstream analysis.
Yes, we accept customer-provided seeds and can also obtain commonly used ecotypes and T-DNA mutants on your behalf.
Yes. We provide a full service, from guide RNA design to screening edited lines and developing homozygous lines.
No, T1 plants are typically hemizygous. We can generate homozygous T2 or T3 lines upon request.
We most commonly use the GV3101 strain for floral dip. We offer a large selection of plant transformation vectors and selection markers (e.g., KanR, HygR, Basta) to suit your specific project requirements.
Yes. In addition to stable transformation, we offer three efficient transient systems: Agrobacterium-mediated leaf transformation, microparticle bombardment, and PEG-mediated protoplast transformation. These are ideal for rapid protein activity evaluation or subcellular localization studies.
Absolutely. While Col-0 is the standard, we have extensive experience with various ecotypes (e.g., Ler, Ws) and can perform transformations using your specific mutant backgrounds.
We offer a highly robust and optimized floral dip platform designed for reliability. For each standard construct, we guarantee the delivery of at least 10–20 independent T1 transgenic lines, providing ample biological replicates for your research. For genome editing projects, we consistently achieve up to 80% CRISPR/Cas9 editing efficiency, ensuring reliable results even for complex target sites.
Yes, we offer a wide range of phenotypic and molecular analysis services.

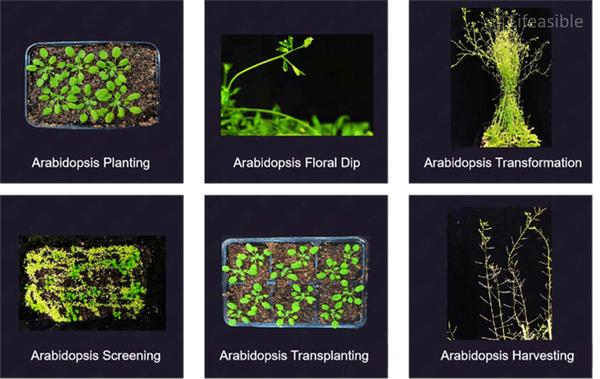
Protocol for Arabidopsis Transformation

Extraction of Soluble Protein from Arabidopsis thaliana Leaves

Unlocking the Basics: What is Genetic Transformation and Why It Matters
CRISPR-Cas9: A Comprehensive Guide to Genome Editing in Plants
Reference