Professional, Customizable Triticum aestivum Transformation Services
Lifeasible specializes in providing high-efficiency wheat transformation services to support your research in functional genomics, molecular breeding, and crop improvement. Our platform offers end-to-end support from codon optimization and vector construction to the delivery of stable transgenic lines, providing tailored solutions for academic researchers and the AgBio industry dedicated to cereal crop advancement.
TARGET GENOTYPES
Fielder, Bobwhite, CB037,
& Elite Commercial Cultivars
TYPICAL YIELD
5–20
Independent T0 Positive Wheat Events
EDITING EFFICIENCY
Up to 70%
High-efficiency CRISPR/Cas9 editing
LEAD TIME
6–9 Months
From bombardment or infection to T1 seeds
Standard Package
Efficiency Focused
Premium Package
Full-Service Custody
Stable transformation is the cornerstone of wheat biotechnology, allowing for the integration of novel traits into the complex hexaploid genome. Our service utilizes both Biolistic Particle Bombardment and Agrobacterium-mediated transformation of immature embryos, refined to overcome the recalcitrance typically associated with cereal regeneration.
Unlike many dicot systems, wheat transformation relies on precise tissue culture-based regeneration. Our optimized media formulations ensure high embryogenic callus induction and green plant regeneration, significantly reducing the frequency of albinism and somaclonal variation.
![]()
Explant Preparation
Isolation of immature embryos from greenhouse-grown donor plants.
![]()
DNA Delivery
Introduction of T-DNA via Agrobacterium or gold particles via bombardment.
![]()
Callus Induction
Specialized selection media to identify transformed cells
![]()
Regeneration
Induction of shoots and roots under controlled light and temperature.
![]()
Hardening & Seed Set
Acclimatization of T0 plants to soil for T1 seed production.
For rapid validation of genetic constructs or protein localization without the long timelines of stable cereal transformation, we offer specialized transient systems. These are ideal for testing sgRNA efficiency or promoter strength before committing to a full-scale stable project.
![]()
DNA Construct Preparation
High-purity plasmid isolation.
![]()
Delivery System
Biolistic bombardment of leaves/coleoptiles or PEG-mediated protoplast fusion.
![]()
Expression Analysis
Reporter detection (LUC/GFP) within 24–72 hours.
| Category | Requirements |
| Sample Type | Healthy donor plants grown under controlled conditions OR isolated immature embryos (IEs) at early milk stage (0.8–1.5 mm diameter, ~12–16 days post anthesis) |
| Sample Amount | Minimum 200–300 immature embryos per construct (recommended 500+ for standard efficiency; typical transformation efficiency 1–5%) |
| Pre-Treatment | Donor plants must be grown in controlled environment chambers (15–20°C day/night) without pesticide/fungicide spraying; spikes harvested at precise developmental stage |
| Storage Conditions | If shipping whole spikes: store at 4°C and process within 24–48 hours; If shipping isolated IEs: maintain on WIM medium at room temperature and ship immediately |
| Shipping | Whole spikes: ship in moist conditions at ambient temperature with ice packs; Isolated IEs: ship in sterile tubes with inoculation medium; Avoid freezing immature embryos |
| Metadata Needed | Cultivar name (Fielder, Bobwhite, Cadenza preferred; commercial varieties may need optimization), growth stage (days post anthesis), greenhouse conditions, ploidy level if known, target gene/construct details, preferred selection markers (bar, hptII, nptII) |
| Vector Information | Complete binary vector map with monocot-optimized promoters (e.g., Ubi, Act1, OsAct1), selection cassette, and reporter genes; Helper plasmid requirements if using pGreen/pSoup system |
Beyond our standard wheat transformation, we offer a suite of advanced genomic and molecular tools to maximize the precision and efficiency of your research:
Custom Vector & Optimization:
Tailored vector construction with monocot-optimized promoters (e.g., Ubi, Act1), selection markers (bar, hptII), and high GC-content codon optimization.
Advanced CRISPR/Cas9 strategies for the hexaploid wheat genome, including triple-genome knockouts, base editing, and prime editing.
Multi-Genome CRISPR Screening:
Specialized bioinformatics and deep sequencing services to precisely track and verify editing events across the A, B, and D sub-genomes.
Transgene-free & Speed Breeding:
Identification of non-GMO edited lines through T1/T2 segregation analysis, combined with speed breeding protocols to accelerate generation turnover.
Elite Cultivar & Library Transformation:
Custom protocol optimization for recalcitrant elite commercial varieties and high-throughput processing for large-scale functional genomics screens.
Vector Construction & Validation
Explant Preparation & Infection/Bombardment
Callus Induction & Selection
Shoot & Root Regeneration
Acclimatization (Hardening)
Seed Maturation (To T1 Stage)
Note: Total project duration typically ranges from 6 to 9 months depending on the genotype and specific project requirements.
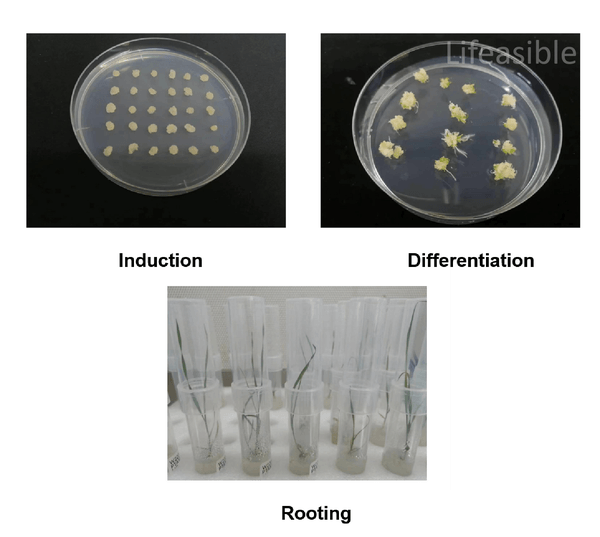
Wheat Transformation for Sweet Protein Expression
Internal project report detailing successful genetic modification of wheat (Triticum aestivum) for sweet protein production. Using Agrobacterium-mediated transformation of immature embryos, this optimized protocol achieved targeted integration of Brazzein and Thaumatin expression cassettes, confirmed via PCR screening and regeneration of fertile T1 plants.

High-Efficiency Wheat Transformation (QuickWheat System)
Recent advancements in hexaploid wheat (Triticum aestivum) transformation demonstrate that the use of morphogenic regulator genes ZmWuschel2 (ZmWUS2) and Zm-Baby Boom (ZmBBM) significantly enhances transformation efficiency by inducing rapid somatic embryogenesis. This approach simplifies conventional tissue culture procedures, reduces genotype dependency, and shortens the overall transformation timeline from ~80 days to ~50 days, enabling efficient generation of high-quality transgenic events across multiple wheat genotypes.
Empowering researchers and agricultural leaders with reliable cereal transformation solutions
"The technical expertise required for hexaploid wheat transformation is significant, and Lifeasible delivered beyond our expectations. Their ability to target all three sub-genomes (A, B, and D) using CRISPR/Cas9 saved our lab months of optimization work."
Dr. Arisawa
Senior Scientist
USA
"From the initial vector design to the delivery of PCR-positive T0 plants, the transparency was excellent. The monthly progress reports allowed us to align our downstream field trials perfectly with their regeneration timeline."
Dr. Henderson
Project Leader
USA
"We struggled with low regeneration rates and albinism in our in-house wheat tissue culture. Switching to Lifeasible's optimized bombardment platform provided us with the high-quality independent lines we needed for our yield-improvement study."
Dr. Petrov
Principal Investigator
Canada
"The team at Lifeasible successfully transformed an elite wheat cultivar that other services refused to touch. Their commitment to project-specific protocol optimization makes them a premier partner for commercial cereal breeding."
Dr. Sterling
Director of R&D
USA
Cereal Expertise
Specialized team focused on the unique requirements of wheat tissue culture.
Genomic Depth
Deep understanding of the hexaploid wheat genome for precise editing design.
Capacity
High-throughput embryo excision and bombardment facilities.
Transparency
Detailed monthly reports on callus health and regeneration progress.
Ready to start your Triticum aestivum transformation project?
Our technical experts are ready to assist you in navigating the complexities of wheat genomics and tissue culture. Whether you are targeting grain quality, disease resistance, or fundamental developmental pathways, we provide the robust platform necessary for your success.
Triticum aestivum, also known as wheat, is one of the most important crops in the world. Due to its enormous nutritional significance and high economic value, wheat is an essential cereal model organism for studying plant growth regulation and crop yield improvement.
Traditionally, wheat has been classified as a recalcitrant crop for genetic manipulation. Unlike model organisms like Arabidopsis, wheat presents unique biological hurdles:
Selecting the right delivery system is critical for balancing speed, cost, and the quality of the resulting transgenic lines.
| Feature | Biolistics (Gene Gun) | Agrobacterium-mediated |
|---|---|---|
| Mechanism | Physical delivery of DNA-coated gold/tungsten particles. | Biological delivery via T-DNA integration. |
| Variety Scope | Highly versatile; less dependent on genotype. | Traditionally restricted, but expanding via specialized strains. |
| Copy Number | Often results in high copy numbers and complex insertions. | Typically yields single-copy and clean integrations. |
| Industry Preference | Used for rapid screening or transient assays. | Preferred for commercial traits due to higher genetic stability. |
Wheat transformation is the bridge between genomic data and agricultural improvement. Its applications span the entire R&D pipeline:
'Fielder' is our standard for high efficiency. We also offer services for 'Bobwhite' and can perform custom optimizations for client-specific elite cultivars.
We perform bioinformatic alignment across the A, B, and D genomes to design sgRNAs that either target all three (for complete knockout) or are specific to a single sub-genome, depending on your goals.
Yes, Southern Blot is available as an add-on service to confirm T-DNA copy number, which is critical for downstream breeding and regulatory compliance.
While 'Fielder' remains the gold standard for wheat transformation, we have extensive experience optimizing protocols for various elite commercial cultivars. For recalcitrant genotypes, we employ specialized strategies—such as adjusting hormone ratios, osmotic pre-treatments, or dual-delivery systems—to minimize genotype dependency and ensure the successful generation of stable T0 positive lines.
Wheat regeneration can occasionally result in chimeras. To ensure reliability, we perform multi-point sampling across different tillers of the same plant for high-sensitivity PCR and qPCR screening. For our Premium Package, we further conduct segregation analysis on T1 seeds to confirm that the transgene is stably integrated and heritable in subsequent generations.
Transformation of Immature Wheat Embryos Using Gene Gun Method

Wheat Embryo Culture Technology

RAPD Analysis of Genetic Diversity for Salt Tolerance in Wheat

Stable vs. Transient Transformation
Reference