Among spice crops, black pepper (Piper nigrum) has long been considered a quintessential recalcitrant species. Despite the continuous growth of its global market, the frequent occurrence of diseases—such as root rot—coupled with a traditional breeding cycle spanning 10 to 20 years, has made the establishment of a molecular improvement system particularly urgent.
However, a core challenge has persisted: although regeneration systems have been established, the efficiency of stable genetic transformation remains low, plant regeneration is difficult, and the process is hard to scale up for industrial application.
On February 28, 2026, Horticulture Research published the latest findings from the team led by Khaled M.A. Amiri at the United Arab Emirates University. The paper, titled "Reducing recalcitrance of black pepper to Agrobacterium-mediated transformation: an efficient way through nucellar apomixis to establish transgenic and genome-edited plants at high frequency and scale-up through bioreactor," presents a novel approach. By utilizing somatic embryos derived from nucellar apomixis as recipient materials, the study systematically optimized transformation conditions, carbon source regulation, and large-scale propagation protocols. Ultimately, the team established a genetic manipulation system that is highly efficient, stable, and scalable.
Under this system, the efficiency of stable Agrobacterium-mediated transformation reached a maximum of 91%; CRISPR/Cas9-mediated editing of the Pds gene reached 89% efficiency; and the survival rate of transgenic plantlets ex vitro reached 99%. Furthermore, under bioreactor conditions, a single system unit could propagate approximately 300 plants within three months, thereby demonstrating significant potential for large-scale production.
One of the primary reasons for the difficulty in genetically modifying black pepper lies in the instability of recipient materials and the limited efficiency of plant regeneration.
The study first established a system utilizing somatic embryos derived from nucellar apomixis. Following the in vitro culture of zygotic embryos, somatic embryos of nucellar origin were observed to emerge from the root-shoot junction. When cultured in SHS3 medium under dark conditions, the formation of somatic embryos was successfully induced in both tested varieties, which subsequently entered a phase of recurrent somatic embryogenesis. Under conditions of 1% sucrose (SHS1), somatic embryos can undergo continuous cyclical proliferation; however, when the sucrose concentration is increased to 3% (SHS3), conditions become more conducive to embryo maturation and subsequent conversion into plantlets. Under illumination, cotyledonary-stage embryos turn green and develop into complete plantlets.
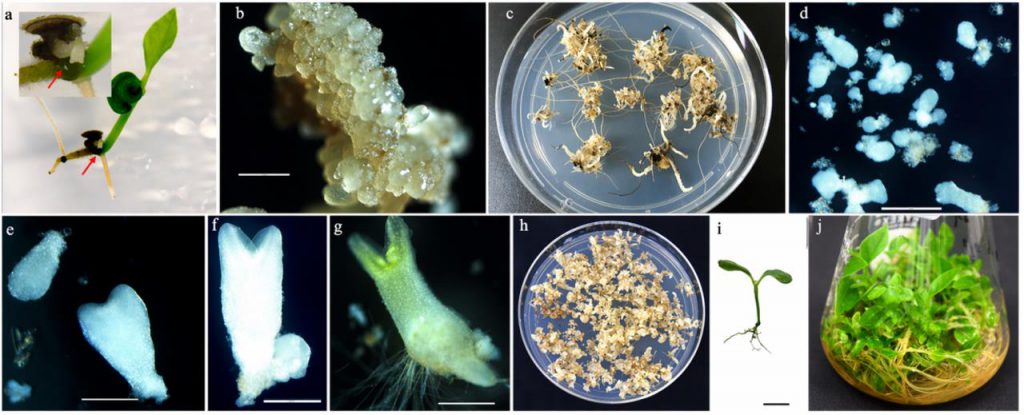
Figure 1. Establishment of a nucellar apomixis-derived somatic embryo system in black pepper. (Sasi, et al. 2026)
The critical significance of this system lies in its ability to ensure a stable and sustainable supply of embryogenic material through cyclical somatic embryogenesis, thereby providing a foundational basis for high-frequency transformation.
Building upon the established regeneration system, this study systematically compared the effects of various carbon sources on somatic embryo conversion and root system development.
The results demonstrated that under conditions of 3% glucose, the conversion rate of early torpedo-stage somatic embryos into plantlets reached 100%, a figure significantly higher than the approximately 83% efficiency observed under 3% sucrose conditions. More importantly, plantlets developed within the glucose-based culture system exhibited robust primary roots and distinct lateral branching, whereas root systems formed under sucrose conditions were comparatively weaker.
This disparity ultimately manifested in the transplant survival rates: plantlets derived from the glucose system achieved a soil survival rate as high as 99%, whereas those from the sucrose system survived at a rate of approximately 53%. Further expression analysis revealed that under conditions of 1% glucose, the expression levels of PnPIN2 and PnDhn1 were at their highest, consistent with the observation that a glucose-enriched environment is more conducive to the healthy development of the root system.
These findings indicate that the optimization of carbon sources not only influences transformation efficiency but also directly determines the structural quality of the plantlets and their subsequent adaptive capacity.
Based on the availability of stable embryogenic material, the study proceeded to systematically optimize key parameters influencing transformation efficiency.
In the absence of sonication, the stable transformation rate stood at a mere 3%; however, when the ultrasound duration was extended to 20 seconds, transformation efficiency rose significantly to 56%. Upon further optimization to 30 seconds, the stable transformation rate reached a peak value of 84%. Extending the duration further to 40 seconds—while resulting in enhanced transient expression—led to a decline in the emergence of embryogenic masses and globular embryos, ultimately causing a slight reduction in the stable transformation rate. Consequently, the study identified 30 seconds as the optimal duration for sonication. Regarding strain selection, AGL1 demonstrated the highest stable transformation efficiency, reaching 91%; EHA105 achieved 84%; and GV3101 reached 65%. Based on a comprehensive assessment of both efficiency and stability, AGL1 was identified as the optimal strain.
Following the optimization of the screening system, transformed tissues remained white or pale yellow and continued to proliferate, while untransformed tissues gradually browned and died. After approximately 50–60 days, cotyledon-stage embryos were obtained and subsequently transferred to a liquid culture system to develop into complete plantlets. The entire process, from infection to soil acclimatization, took approximately 90 days.
The study also established an Agrobacterium rhizogenes-mediated hairy root system. Following infection, hairy roots expressing either GFP or RFP were successfully generated.
In the composite plant system, the transformation efficiency for hardened plantlets derived from somatic embryos was approximately 70%, whereas the efficiency for direct stem cuttings was about 55%. Transformed roots could be clearly distinguished from untransformed roots using fluorescence imaging. This system provides a technical pathway for validating the function of root-specific disease-resistance genes.
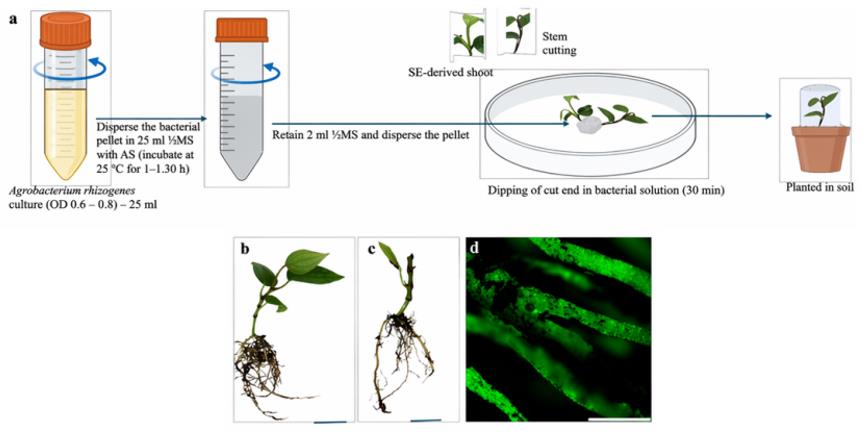
Figure 2. Generation and validation of composite plants in black pepper. (Sasi, et al. 2026)
To validate the editing capability of the system, CRISPR/Cas9 editing was performed using the Pds gene as the target. Approximately 89% of the infected embryogenic mass-derived embryos exhibited a characteristic albino phenotype. Sequencing results revealed the occurrence of insertion or deletion mutations at the target site, with some mutations resulting in the introduction of premature stop codons. All edited plantlets displayed the albino phenotype, accompanied by stunted growth and reduced leaf size.
The study utilized a Temporary Immersion Bioreactor (TIB) system for large-scale propagation. Compared to suspension culture, the proliferation rate of embryogenic tissues in the TIB system was approximately twofold higher. Through an optimized protocol involving daily immersion for 5–10 minutes, healthy plantlets were consistently produced. A single TIB system was capable of producing an average of approximately 300 plantlets within a three-month period, maintaining a transplant survival rate of 99%.
In summary, through the establishment and systematic optimization of a somatic embryogenesis system based on nucellar apomixis, this study successfully overcame the bottlenecks in the genetic modification of black pepper. It thereby constructed a complete technological closed loop, spanning from high-frequency transformation and CRISPR editing to large-scale propagation. This system not only provides a practical pathway for the molecular breeding of black pepper but also offers a valuable paradigm for other perennial crops that are typically recalcitrant to genetic transformation.
| Cat# | Product Name | Size |
| ACC-100 | GV3101 Chemically Competent Cell | 100μL/tube |
| ACC-103 | EHA105 Chemically Competent Cell | 100μL/tube |
| ACC-105 | AGL1 Chemically Competent Cell | 100μL/tube |
| ACC-107 | LBA4404 Chemically Competent Cell | 100μL/tube |
| ACC-108 | EHA101 Chemically Competent Cell | 100μL/tube |
| ACC-117 | Ar.Qual Chemically Competent Cell | 100μL/tube |
| ACC-118 | MSU440 Chemically Competent Cell | 100μL/tube |
| ACC-119 | C58C1 Chemically Competent Cell | 100μL/tube |
| ACC-121 | K599 Chemically Competent Cell | 100μL/tube |
| ACC-122 | Ar.A4 Electroporation Competent Cell | 50μL/tube |